Part I: The anaphylactic patient
This post is the first of a planned three-part series. In this introduction, I realize that my trauma bay does not have an awake intubation kit. This article, however, is not about awake intubation itself, as there are already many articles out there. Instead, it is about the logistical challenges of not having a ready kit, and a call for input from the social media community. – Daniel
***
Having napped poorly before a night shift, I am caffeinating myself when the trauma nurse tells me about a patient who is having an allergic reaction and needs to be seen right away.
Clearing the cobwebs from my eyes, I go to the trauma bay, where the paramedics are rolling in an elderly woman named Agnes. Agnes is sitting upright on the stretcher, her chin cocked into a sniffing position, and mouth-breathing at 50 breaths per minute.
Her tongue is red, enormous, and wet with saliva. She can hardly talk. From her husband and her medication list, we learn that Agnes takes a few cardiac medications, but there have been no recent additions or changes. One of these medications is an angiotensin-converting enzyme (ACE) inhibitor, although she has been on the medication for years without incident.
We throw the kitchen sink of anaphylactic treatments at Agnes: epinephrine, Benadryl, famotidine—we even order C1 esterase inhibitor. Nothing seems to quell the inflammation and an airway intervention looms.
Given the pathologic, inflamed airway, a standard rapid-sequence intubation is risky, as neuromuscular blockade could precipitate a disastrous closed airway after protective reflexes are abolished [1]. Instead, we discuss performing an “awake look” to visualize the anatomy first with protective reflexes present, before attempting to pass the endotracheal tube through the vocal cords.
We predict a difficult airway; in addition to the swollen anatomy, Agnes also has a short, arthritic neck. We call our anesthesia colleagues to help. In the meantime, we landmark and prep the neck and open a cricothyrotomy kit by the bedside.
As we wait for our anesthesia colleagues, we begin to apply topical anesthetic to the upper airway by nebulizing lidocaine. Someone tells us our hospital has stopped carrying 4% lidocaine, so we nebulize a 2% solution. We also administer glycopyrrolate to decrease secretions.
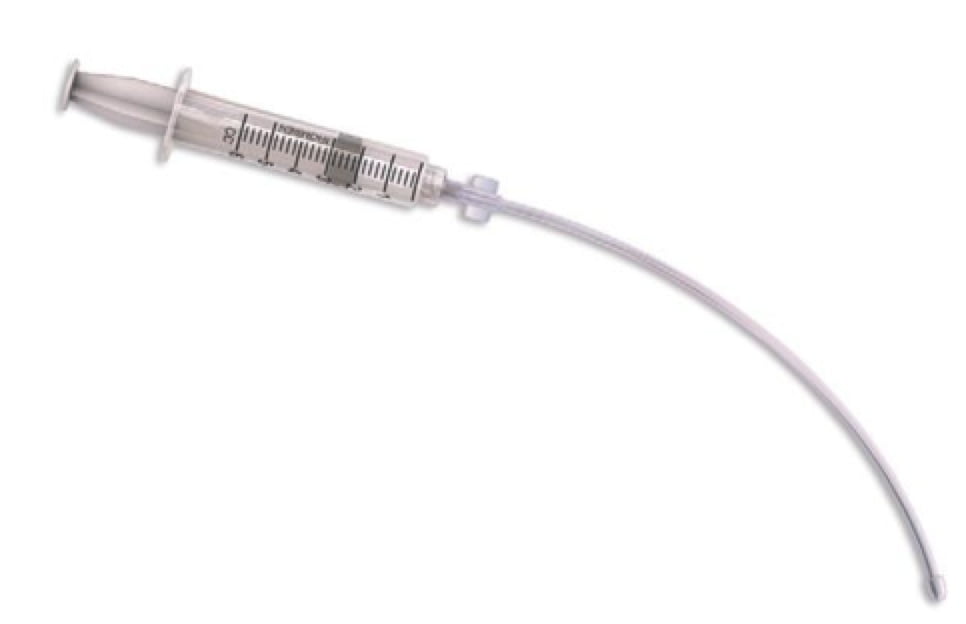
When the anesthesia attending arrives, he agrees with our plan to do an awake intubation, and wants to topicalize down to the vocal cords. He asks for a specific mucosal atomization device (MADgic), but we do not carry it in the Emergency Department. We scrounge up a few different devices in the department, but the anesthesiologist calls the operating room for his preferred device. It takes what seems like forever for the atomizer to arrive. In the meantime, our patient is stable but looking increasingly uncomfortable.
Figure 1. This laryngo-tracheal mucosal atomization device attaches to a standard syringe [1].
Fortunately, my case has an uneventful ending. The atomizer arrived, we finished topicalizing the airway, and we are able to get a Grade 1 view with our video laryngoscope. (Finding out the patient wore dentures was a bonus.) We passed through the cords easily, and then sedated and paralyzed the patient. We congratulated one another on a job well done and a disaster averted.
Afterward, one of the emergency physicians observed that we re-invent the wheel every time we do an awake intubation, assembling an ad hoc collection of drugs and devices. I wondered if the long delays associated with not having a pre-assembled kit could lead to patient harm in the future.
My question to the social media community is this:
Over the next couple of months, my colleague, Jared Baylis, and I will be assembling an awake intubation kit for our Emergency Department.
We would like to invite you all to help us. Please join and share in the spirit of #FOAMed in the comments below.
Questions to the Audience:
1) Do you have a kit in your shop? If so, what are your practical, trusty go-to ingredients?
2) What does your ideal awake intubation kit look like?
In particular, we are interested in hearing practical input into the various approaches to topical anesthetic and whether choice is situation-dependent.
In part II of this series, Jared and I will be using your ideas and meeting with some local airway experts to gather their insight into their practice preferences. In part III, we will be assembling the kit and posting it online for viewing, critique and commentary.
References:
- Schwartz, R.B., Shepherd, G (2014). Pharmacologic adjuncts to intubation. In: Roberts, J.R., Custalow, C.B., Thomsen, T.W., & Hedges, J.R. Roberts and Hedges’ clinical procedures in emergency medicine. 6th ed. Elsevier Saunders, Philadelphia, PA. 118-119.
Reviewing with the Staff
This post has been reviewed by Dr. Alim Pardhan (@alimpardhan), assistant professor at McMaster University and clinical site lead at Hamilton General Hospital.
This is an excellent example of how frustration, or a cardinal event, can lead to innovation and process changes in the clinical environment. Making changes in the clinical environment can sometimes be frustrating, but can also be rewarding when everything finally comes together. The process that you follow to make changes is variable, but having a process can be helpful in ensuring that everything gets done.
There are a number of tools that exist to help with managing these types of changes, most of which are available online. Here, however, are some practical tips:
- Ensure that you have a clear target. What are you trying to achieve and why is it important? Identify what will success look like – not only that you “have a kit” but also what are the outcomes you hope will be managed?
- Identify all your stakeholders. For this type of kit, this includes knowing who will be using it (e.g. ED, Anesthesia, Trauma + associated learners), who will be supporting it (e.g., RN, RT), who will be stocking it, and who will be paying for it. What are the needs of these stakeholders, and will they need training? When should they be consulted?
- Don’t reinvent the wheel. Solicit input from others – both locally and elsewhere – for input. Someone else has likely already done this, and knowing how they solved the problem can help you avoid unnecessary work.
- Make sure you have a process to manage all the input you get. Who will decide what goes into the kit, and how will they decide? Ideally do this before you start collecting and analysing input. One way to do this is to divide the input you receive into categories like ‘must have,’ ‘nice to have,’ and ‘don’t need’.
- Make sure you have a process to debrief after your solution is used so that you can troubleshoot any issues that come up.
- Have a sustainability plan. How will you make sure it gets restocked after it gets used?
Happy Building!