It has been a slower than usual night shift, and you have just led a resuscitation for a COVID-19 patient in your ED. Your ED has experienced several policy changes in recent weeks, from new standards for the use of personal protective equipment to policies for mitigating risk during aerosol-generating medical procedures to guidelines on how to best communicate between resuscitation and support teams. You wonder how your department can implement multiple changes so quickly, while at the same time ensuring that all the staff are up to date on these changes. You feel anxious about all these changes and want to practice these new protocols and procedures before engaging in real-life protected codes and resuscitations.
Welcome to our second special HiQuiPs post on innovation during the COVID-19 pandemic. In our first post we discussed adaptations of general quality improvement (QI) principles during a time of pandemic planning that is full of change. We continue by highlighting the utility of blending QI and simulation techniques, which can allow departments to rapidly identify latent safety threats (LSTs), and adapt during a particularly high-risk time for both patients and providers: the resuscitation (or, in COVID-19 terms, the protected resuscitation/protected code blue).
Adverse Events and Latent Conditions
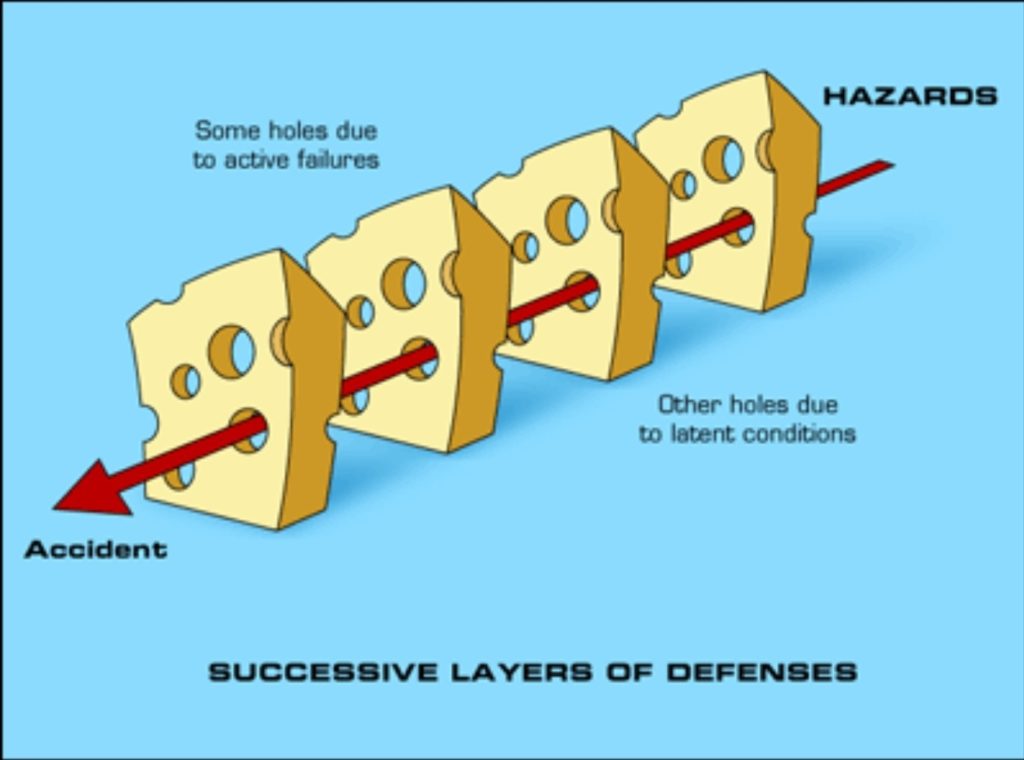
Protected resuscitations are high-risk, extremely variable acute presentations that place large demands on the ED environment and its members. Adverse events in this environment may carry significant weight for both patients and providers. These adverse events may arise from either active failures or latent conditions.1 Active failures are unsafe actions taken by people and include mistakes, slips, fumbles, and procedural violations.2 Latent conditions, by contrast, are a series of individually innocuous conditions that, when spontaneously aligned, lead to adverse effects (Figure 1). By identifying the holes in each individual slice of Swiss cheese, we can prevent such latent adverse events from occurring.
High Reliability Organizations
High Reliability Organizations (HROs) are systems designed to operate in hazardous conditions (for example, aviation, military and nuclear power) but to do so with high levels of safety.3 These organizations have specific characteristics, most notably an intense preoccupation with preventing failure.4 Obsession with the prevention of failure prevents complacency and heightens vigilance for errors. In the hazardous setting of ED protected resuscitations, there are a few potential approaches that organizations can use to work towards a highly reliable, safe culture. One such approach is the use of in situ simulation (ISS).
Rapid-Cycle In Situ Simulation
A novel rapid cycle ISS program was developed at Toronto General and Toronto Western Hospitals for this purpose.5 This enabled an interdepartmental and interprofessional team to uncover and address departmental, organizational, and systemic gaps that presented LSTs to both patients and providers quickly, in order to reach both educational and institutional saturation.
ISS exercises are simulation exercises conducted in the clinical workspace by care providers who are on clinical duty, using equipment and resources in that workspace.6 ISS can identify LSTs to patients and providers, augment workflows in resuscitations, improve communication amongst teams, and provide an opportunity for deliberate practice of critical procedures and skills.7
The Benefits of Rapid-Cycle In Situ Simulation
Rapid cycle simulation has previously been described in medical education as a method of achieving mastery of resuscitation principles.8 By incorporating ISS into the PDSA rapid cycling method, EDs can a hybrid “rapid-cycle ISS program” to prepare for an emerging respiratory pathogen. There are examples of ISS being used in an iterative way to find solutions to LSTs over months to years.9,10 However, this hybrid program accelerates the process, performing simulation cycles over days to weeks. The rapid-cycle design allows for iteration of multiple improvement cycles within and in between sessions, allowing teams to innovate and adapt quickly during a global pandemic. It circumvents the challenge in pandemic planning of developing a process and revisiting it weeks later to gather feedback; it allows for rapid improvements to be implemented before being enacted in clinical care. This is achieved through simulation in a rapid timeframe, but also by brining all institutional, departmental and front-line stakeholders around the same “table” (or, in this case, the same mannequin).
An Example of a Rapid-Cycle ISS Program
Our rapid-cycle ISS program included five ISS sessions in rapid succession over six weeks. It included all relevant stakeholders, including ED physicians, nurses and respiratory therapists. LSTs identified included personal protective equipment (PPE) issues, communication, and environmental challenges. Every session included an opportunity to find a problem and to suggest a solution that could be tested in the next session (Figure 2).
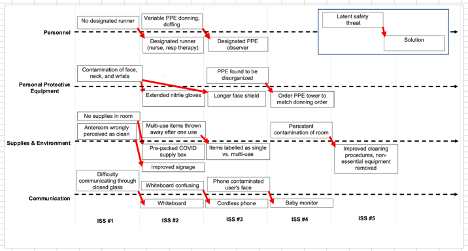
LSTs that were identified by simulation exercises were divided into four categories: personnel, PPE, supplies and environment, and communication. Each time an LST was identified (box above hashed line), a solution was generated to be tested in a subsequent in-situ simulation exercise (box below hashed line). This process was iteratively done until no new LST was identified (i.e. saturation was achieved).
Summary
Protected resuscitations present high-risk situations that are also evolving as best evidence practice emerge during the evolution of the pandemic. Rapid-cycling ISS provides a synchronous blend of QI and simulation that uncovers LSTs, decreases uncertainty, and prepares for failures in an HRO. The approach also allows various levels of stakeholders – from policy makers to front line personnel – to contribute to the process and optimize preparation for the next sick patient.
Thank you for tuning in to our special post on Rapid-cycle ISS! Join us next time as we go back to the basics and discuss the preparation of QI manuscripts to help you disseminate your work!
This post was copyedited by Matthew Hacker Teper (@matthackerteper)
**UPDATE (November 2020): The HiQuiPs Team is looking for your feedback! Please take 1 minute to answer these three questions – we appreciate the support!**
- 1.Reason J. Human error: models and management. West J Med. 2000;172(6):393-396. doi:10.1136/ewjm.172.6.393
- 2.Reason J. Human Error. Cambridge University Press; 1990.
- 3.Carroll J, Rudolph J. Design of high reliability organizations in health care. Qual Saf Health Care. 2006;15 Suppl 1:i4-9. doi:10.1136/qshc.2005.015867
- 4.High Reliability. Agency for Healthcare Research and Quality. Accessed April 17, 2020. https://psnet.ahrq.gov/primer/high-reliability
- 5.Dharamsi A, Hayman K, Yi S, et al. Enhancing departmental preparedness for COVID-19 using rapid-cycle in-situ simulation. J Hosp Infect. 2020;105(4):604-607. doi:10.1016/j.jhin.2020.06.020
- 6.Petrosoniak A, Auerbach M, Wong A, Hicks C. In situ simulation in emergency medicine: Moving beyond the simulation lab. Emerg Med Australas. 2017;29(1):83-88. doi:10.1111/1742-6723.12705
- 7.Cheng A, Grant V, Auerbach M. Using simulation to improve patient safety: dawn of a new era. JAMA Pediatr. 2015;169(5):419-420. doi:10.1001/jamapediatrics.2014.3817
- 8.Taras J, Everett T. Rapid Cycle Deliberate Practice in Medical Education – a Systematic Review. Cureus. 2017;9(4):e1180. doi:10.7759/cureus.1180
- 9.Yager P, Collins C, Blais C, et al. Quality improvement utilizing in-situ simulation for a dual-hospital pediatric code response team. Int J Pediatr Otorhinolaryngol. 2016;88:42-46. doi:10.1016/j.ijporl.2016.06.026
- 10.Barbeito A, Bonifacio A, Holtschneider M, et al. In situ simulated cardiac arrest exercises to detect system vulnerabilities. Simul Healthc. 2015;10(3):154-162. doi:10.1097/SIH.0000000000000087



