This episode of CRACKCast covers Rosen’s Chapter 79, Dysrhythmias. This encompasses a variety of pathologies with a spectrum of management from immediate intervention to watchful waiting. Learn the essentials of the conduction pathways, where things can go wrong, and what rhythms to watch for – here.
Shownotes – PDF Here
[bg_faq_start]Rosens in Perspective
“In nonpacemaker cells, additional depolarization from a second electrical stimulus is not possible when the membrane potential is more positive than −60 mV.”
Two main types of myocytes
- Non-pacemaker cells
- Function depends on Na, K, and Ca and resting potential of -90mV
- K naturally flows out of the cell, and ATP works with Mg+ as a cofactor to transport Na+ outside the cell using K+=ATPase pump
- **non-pacemaker cells can undergo depolarization in pathologic conditions (e.g. ischemia)**
- Pacemaker cells:
- Exist in SA and AV node ; surface of AV valves and His-Purkinje system
“Dysrhythmias are classified according to their electrophysiologic origin, ECG appearance, and underlying ventricular rate. Although overlap exists, the following categorization is useful:”
- Bradycardias
- Extrasystoles
- Narrow-complex (QRS less than 0.12 second) tachycardias (regular and irregular)
- Wide-complex (QRS 0.12 second or greater) tachycardias (regular and irregular)
Classically, the approach to any specific dysrhythmias is defined on the basis of the clinical stability. Unstable patients demonstrate evidence of severe or multiple end-organ features of hypoperfusion, such as:
- Altered sensorium
- Respiratory distress
- Hypotension
- Syncope
- Chest pain suggestive of myocardial ischemia.
Stable patients may be asymptomatic or have mild symptoms, such as light-headedness, dyspnea on exertion, palpitations, or mild anxiety. In practice, clinical stability is a continuum; in the absence of profound altered sensorium or hypotension, a clear line distinguishing stable and unstable patients is often not present.
It is important to consider whether a dysrhythmia is the cause or an effect of a clinical presentation; for example, rapid atrial fibrillation may be causing hypotension or resulting from profound volume depletion. Failure to consider the clinical situation can lead to an inappropriate focus on diagnosing and treating the rhythm to the detriment of the patient. With recognition of this need to incorporate the overall clinical picture, treatment of those with a dysrhythmia and clear instability is empirical and assumes the rhythm is the cause, whereas stable patients can be approached in a more systematic and thoughtful manner to identify the cause and choose the most appropriate therapy.”
[1] What is the blood supply of the following parts of the conduction system: SA node, AV node
Anatomy and conduction :
- SA – impulse generating @ RA and SVC
- 55% of people: blood flow from RCA
- 45% ppl = left circumflex
- 60-90 bpm
- Hypothermia and vagal stimulation slows the rate
- Hyperthermia and sympathetic stimulation increases rate
- AV node: 45-60 bpm
- 90% of people = blood flow from RCA (known as RIGHT DOMINANT)
- Abnormal accessory pathways bypass the normal slowing action of the AV node leading to “pre-excitation syndromes”
- The RBB and LSAB = usually LAD supply
- LPIB = usually LCx or RCA
- Short PR interval = impulse generated from lower atria or with an accessory pathway
- PR prolongation = SA or nodal conduction disease
- Three main bundle branches — > then His-Purkinje system
- QT interval = ventricular depolarization to repolarization
[2] What are the three pathophysiologic mechanisms for dysrhythmias?
Mechanisms for dysrhythmia formation
1) Enhanced automaticity:
- Ectopic focus in non-pacemaker cells OR lower threshold in pacemaker cells:
- MI, digitalis toxicity, drugs, electrolyte disturbances
2) Triggered activity
> early or delayed after depolarizations
- Torsades de pointes (TdP); intracellular Ca+ overload, post MI reperfusion
3) Re-entry
> due to abnormal conduction: creating circus movement
- two pathways, one with a longer refractory period so conduction goes down the faster pathway.
- Usually the cause of VT and SVT
[3] What causes & how does AV nodal reentry tachycardia occur?
“For a re-entry mechanism, two alternate pathways for conduction must be present and one path must have a longer refractory period. The unequal responsiveness of the limbs creates a functional unidirectional block such that when the impulse exits one limb, it may then reenter the other in retrograde fashion. The cycle is then repeated, creating a self-sustaining or “circus movement” tachycardia that can appear orderly or disorderly (i.e., fibrillatory).” – Rosen’s 7th Edition
[4] List the classes of antidysrhythmics. Describe their mechanism of action and usual uses, as well as an example of each
Classification of Antidysrhythmics : **all have some prodysrhythmic effects**
| Class I: fast Na+ channel (slow conduction atria, AV, His-P, etc.) Other drugs:
| Class II: beta-adrenergic ANTagonists: suppress SA automaticity and slow AV node conduction | Class III: prolong repolarization and refractory period (K+ channels)
Other drugs:
| Class IV: Ca+ channel blockers (slow AV node conduction) for SVT’s
| Other |
| IA: >PROCAINAMIDE< 20-30 mg/min (total dose 12-20 mg) or ~1g. Risk of Htn > propranolol | ● for Vent rate cntrl ● AVnRT Metoprolol Esmolol
| trxt of atrial and ventricular dysrhythmias | Diltiazem 0.2-0.3 mg/kg bolus minor vasodilation effects, are reversed with 1g Ca | MgSO4: for TORSADES and VT |
| IB: >LIDOCAINE<
| B1: heart +stim: ^ HR , ^ ectopy, ^contractility -antagonism: dec. HR, ectopy, and contractility
| Amiodarone: multiple class effects, long half life Sotalol. | Digitalis: Na/K ATPase blocker with multiple effects (see tox) used in SVT, Afib/flutter 0.25.0.5 mg IV interactions when electrolytes are off! other Na/K atpase:
| |
| IC: >flecainide, PROPAFENONE< only oral use >15% increased vent. dysrhythmias | B2: lungs, vasculature +stim: dec. tone / relaxation -antagonism: increased tone | adverse effects: Hotn, bradyC, dec. contractility, corneal deposits, lung/thyroid toxic, drug interactions | Adenosine:
|
“The subclasses IA, IB, and IC are distinguished on the basis of differential effects on depolarization, repolarization, and conduction.” – Rosens 7th Edition
[5] How does digoxin work as an antidysrhythmic? When is it used?
“Digitalis compounds have a variety of effects on myocardial cells. Digoxin inhibits the ATP-dependent Na+-K+ exchange pump, increasing intracellular Na+ concentrations and decreasing intracellular K+ concentrations. The resultant increase in intracellular Ca2+ concentration accounts for the positive inotropic effects of digitalis. Digoxin can behave as an excitant, a depressant, or both. Excitant effects of digoxin have to do with enhanced automaticity and triggered activity, particularly at high therapeutic or toxic doses. At the same time, digoxin slows AV node conduction at therapeutic doses via lengthening of the refractory period.”
“Digoxin (0.25-0.5 mg IV) can control the ventricular rate in patients with SVTs, including atrial fibrillation and atrial flutter. Because of its delayed onset of action and narrow therapeutic window, however, digitalis is not a first-line agent for emergency therapy. It is not true that digoxin promotes conversion to a sinus rhythm any more than other rate-controlling agents.” – Rosens 7th Edition
[6] List underlying etiologies of sick sinus syndrome.
Umbrella term for a group of dysrhythmias from disease of the sinus node and its surrounding tissues:
- Sinus bradycardia, sinus arrest, and SA exit block.
- A variant of SSS: bradycardia-tachycardia syndrome:
- These bradydysrhythmias alternating with a tachydysrhythmia, typically atrial fibrillation.
“SSS is most common in elderly adults, a result of fibrotic degeneration of the cardiac conduction system. It is also associated with cardiomyopathies, connective tissue diseases, and certain drugs.”
In the acute setting, SSS is suspected when an elderly patient has syncope and the ECG demonstrates sinus impulse abnormalities. Long-term management often requires permanent pacemaker placement for symptomatic bradycardia, in addition to any conventional therapy for atrial fibrillation.”
Keep them slow…..even though they need a pacemaker!
[7] What are three ECG presentations of sick sinus syndrome?
Think about it in any elderly patient with syncope!
- Tachy-brady syndrome
- A fib with episodes of incomplete sinus block
- Complete sinus block
[8] Define 1° heart block. List three causes.
For a quick recap of bradycardias:
BRADYCARDIAS & BLOCKS
<60 BPM, failure of the SA node = escape rhythm is from the ectopic pacemaker.
SINUS BRADYCARDIA
- Normal P; fixed P-P interval
- Conditions:
- Vagal tone; athlete; hypothermia; hypoxia; interior MI; drug effects; sick sinus
- Treatment
- Atropine 0.5 (max 3mg), epi infusion; pacing
SINUS DYSRHYTHMIA
- Variable P-P interval
SINUS ARREST WITH SA EXIT BLOCK
- Missing P waves on ECG
- Sinus block = no impulse conducted from the SA node
- Sinus arrest = no impulse generated
- Can be benign (vagal tone) or pathologic
FIRST degree AV block
- This disease is “lower down” on the pathway than SSS or the sinus dysrhythmias
“This can occur at the level of the atria, the AV node (most common), or the His-Purkinje system”
- Prolonged conduction, but all impulses conducted
- PR > 0.2 sec, normal QRS
Etiologies:
- Can be normal in up to 2% of people
- Ischemic heart disease
- Athletes
[9] Differentiate between the two types of 2° heart block with respect to etiology, ECG appearance and management.
Second degree
- One or more impulses don’t reach the ventricle
- Type I
- wenckebach or mobitz 1
- progressive lengthening of PR until a beat is dropped
- “grouped beats”
- increased vagal tone conditions
- Type II
- block BELOW the AV node
- atropine WORSENS*** this block
- Type I
[10] What is ‘high grade’ heart block? How is it managed?
“a sinus rhythm at a rate of 70 P waves per minute with a 2 : 1 ratio (a ventricular rate of 35 beats/min) represents profound AV block and dysfunction. The term high-grade second-degree block is best applied to conduction disturbances that prevent physiologic ventricular response rates and not solely to higher conduction ratios.”
E.g. 2:1 conduction with atrial flutter going at 300 bpm, means the ventricular rate is 150…which is normal, but in the case above, the impulse is being SLOWED down much more than normal (AV node generates impulses normally at a rate of 45-60 bpm)…
Managed with pacing, because atropine is normally ineffective.
[11] Describe the difference between AV dissociation & 3° AV block.
Third degree AV block:
- Absent conduction of ALL atrial impulses
- Slow escape rhythm
AV dissociation:
- Hallmark of complete heart block
- No electrocardiographic relationship between A/V activity
- R-R interval is longer than the P-P interval
- “Conversely, the presence of AV dissociation with an R-R interval that is shorter than the P-P interval (e.g., as occurs with accelerated junctional rhythms and VTs) does not imply third-degree heart block.”
- **watch for Afib is accompanied by complete heart block. “regularized atrial fibrillation” is associated with digitalis toxicity
FROM LIFE IN THE FAST LANE:
“Complete heart block should not be confused with:
- High grade AV block: A type of severe second degree heart block with a very slow ventricular rate but still some evidence of occasional AV conduction.
- AV dissociation: This term indicates only the occurrence of independent atrial and ventricular contractions and may be caused by entities other than complete heart block (e.g. “interference-dissociation” due to the presence of a ventricular rhythm such as AIVR or VT).”
[12] Compare PAC’s to PVC’s.
- PAC’s
- Abnormal P wave earlier in the cycle than it should appear
- Benign
- may herald myocardial irritability, catecholamine excess, ischemia, heart failure, hyperthyroidism, metabolic derangement
- PVC’s
- Wide QRS with no preceding P wave.
- can create a variety of pauses: compensatory pause, noncompensatory pause
- most with a LBBB appearance are from a focus in the RV
- Multifocal PVC’s
- PVCs coming from more than one source!
- Benign
- catecholamine excess, pain, anxiety, stimulant use
- Pathologic:
- MI, electrolytes, medication toxicity
- bigeminy, trigeminy
- Trxt: reassurance, low dose BB, lidocaine (if developing VT)
What is a compensatory pause? Is it more common with premature atrial or premature ventricular complexes, and why?
“Most PVCs capture the AV node, making it refractory to the next arriving atrial impulse. Because retrograde conduction of a PVC rarely extends far enough to capture and reset the SA node, however, atrial impulses continue to arrive at the AV node at the intrinsic sinus rate. As a result, the R-R interval surrounding a PVC ends up being equal to twice the intrinsic R-R interval length (see Fig. 79-19), a phenomenon known as a compensatory pause. Rarely, a PVC will capture the SA node, resulting in a noncompensatory pause, or will fail to capture the AV node, leaving the underlying rhythm completely unaffected (a so-called interpolated PVC; Fig. 79-20).” – Rosen’s 7th Ed.
[13] Give a differential diagnosis of irregularly irregular tachycardia.
First, a note on narrow complex tachycardias
NARROW COMPLEX TACHYCARDIA
rates>150 make P waves tough to find (sinus Tach vs. Aflutter)
- SINUS TACHYCARDIA
- regular narrow tachy, variability
- <170 in adults ; <225 in infants and children
- treat the problem, NOT the tachycardia
- causes: exogenous/endogenous sympathetic excess
Irregularly Irregular Tachycardias
- M.A.T.
- three or more distinct P waves.
- varying PR and P-P intervals
- associated with COPD
- Trxt: find the underlying cause (Low 02, drugs, electrolytes). Cardioversion doesn’t help.
- ***CAUSES OF irregular/chaotic rhythms:
- 1) A fib
- 2) Atrial tachycardia/flutter with variable conduction
- 3) M.A.T.
- 4) Multiple extrasystoles
- 5) Wandering atrial pacemaker
- 6) Parasystole
[14] Define pre-excitation and list the 3 features of classic WPW
“Pre-excitation refers to depolarization of the ventricular myocardium by circumvention of the AV node via an accessory pathway or bypass tract linking the atria to the ventricles. Accessory pathways lend themselves to re-entry tachycardias and very rapid ventricular rates. Wolff-Parkinson-White (WPW) syndrome is the classic accessory pathway syndrome, characterized by paroxysmal tachycardia and the following three resting ECG features.” – Rosen’s 7th Ed.
- Paroxysmal tachycardia with
- 1) short PR (<0.12)
- 2) QRS > -0.10
- 3) slurred upstroke to QRS (delta wave)
“Patients with WPW and other pre-excitation syndromes often have one or more of the classic features missing on the surface ECG. Also, some patients with WPW-like patterns on their resting ECG never develop symptomatic tachycardia.”
[15] Explain the concept of antidromic and orthodromic conduction with respect to WPW. Which patients with WPW should not receive AV node blockers? Why?
Orthodromic AV- re-entry tachycardia (more common > 70% of cases of WPW)
- AV node used for anterograde conduction and the accessory pathway used for retrograde conduction
- ****NARROW QRS**** is produced
- Presence of A fib. could also occur
- HARD to distinguish from AvNRT
“Orthodromic AV-re-entrant tachycardia is indistinguishable clinically from AVNRT unless the presence of an accessory pathway is known and is treated in the same way, with vagal maneuvers and adenosine as first-line therapy, followed by calcium channel blockers and beta-blockers as second-line agents.”
Antidromic AV-re-entry tachycardia (RARE)
- Accessory pathway is being used as the anterograde limb and the AV node as the retrograde limb,
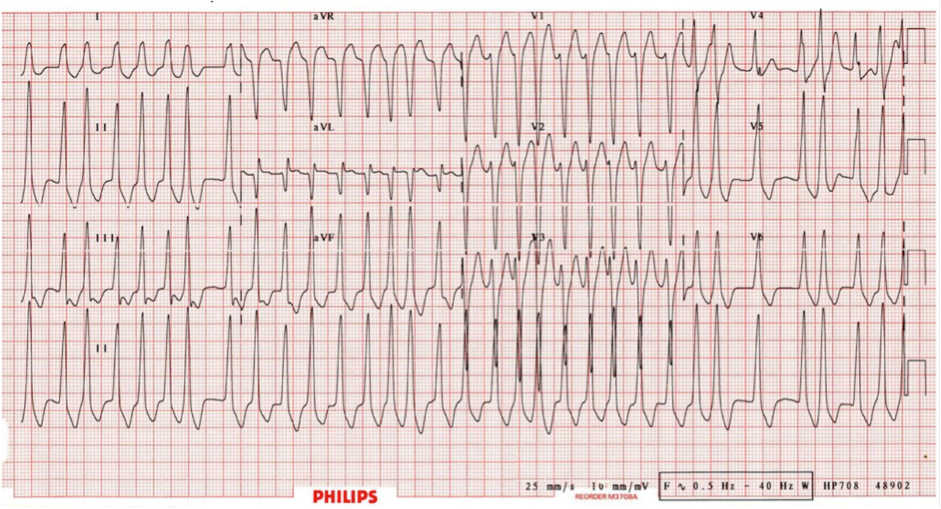
- ***WIDE QRS***
- rapid rate>200
- ”classic nightmare ECG: A fib, with a rate of 200 and a wide QRS”
Copied from: https://emergencymedicinecases.com/episode-20-atrial-fibrillation/
- AV Blocking agents contraindicated
Treatment 1st line: Procainamide, 2nd line: Amiodarone; then cardioversion if unstable.
“Likewise, with a very rapid irregular tachycardia (i.e., atrial fibrillation with a ventricular rate exceeding 200) accompanied by a wide-complex QRS, the likelihood of accessory pathway conduction should be considered and nodal blocking agents again withheld. Procainamide is recommended any time an accessory pathway with wide QRS complexes or very rapid rates exists, with amiodarone as a secondary choice despite theoretic concerns about its nodal blocking properties. Electrical cardioversion (100-200 J) is indicated for ventricular rates greater than 250 beats/min, clinical deterioration, or failure of pharmacologic therapy.”- Rosen’s 7th Ed.
[16] List 10 causes of PVC and VT
- WCT are ANY dysrhythmia with QRS > 0.12 sec.
- may originate in ventricles or above the AV node with aberrant conduction.
- any new Wide TachyC = VT until proven otherwise
- treatment
- unstable: cardioversion
- stable: procainamide or amiodarone
- adenosine may be attempted IF:
- nor irregularity
- ventricular rate <250
- adenosine may be attempted IF:
[17] How do you differentiate between SVT with aberrant conduction & ventricular tachycardia?
Bottom line, when in doubt treat it like VT!
- V-TACH
- see table 79-5
- Age>50, hx of any heart disease
- QRS>0.14sec
- Originates below or at the HIS bundle
- non sustained: < 30sec
- Monomorphic
- Rates 150-200
- Polymorphic VT =
- Bidirectional Vtach = ***digitalis toxicity*****
- Torsades de pointe
- DX:
- “brugada four step approach” see pg. 1058 (very impractical..92% sensitivity.). ALL questions: any questions answered “yes” suggests VT
- ?No RS complexes in the chest leads
- ?RS duration (beg. of R to deepest S) > 100 msec
- ?AV dissociation
- ?VT morphologic criteria:
- notched S in V1
- QR or QS in V6
- “brugada four step approach” see pg. 1058 (very impractical..92% sensitivity.). ALL questions: any questions answered “yes” suggests VT
- Griffith approach. 92% sens
- No classic RBBB/LBBB morphology
- presence of AV dissociation (i.e. p-waves are not all preceeding the Ventricular depolarization)
- Trxt:
- Stable
- Amiodarone 3-5 mg/IV or lidocaine 1-1.5 mg/kg IV
- Procainamide 30-50 mg/kg IV is second line
- Unstable = Shock!
- Stable
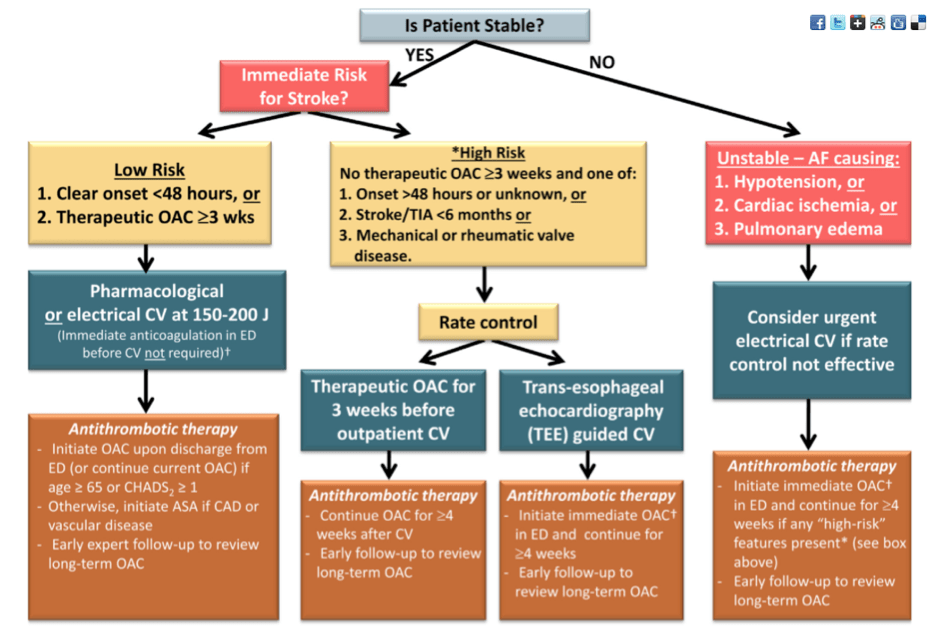
[18] List 10 causes of atrial fibrillation.
[19] Describe the management of AFIB, including a discussion about the CHADS2 score and long term stroke risk.
…..This is where Rosen’s lets us down. Some of the information here is not standard of care currently. We’d suggest you check out:
But for the sake of a quick overview, let’s cover some of the details…
- Afib = electrical chaos, no coordinated atrial contractions (less filling, less Stroke Vol)
- Classified as: Paroxysmal / persistent / permanent
- if the conducted rate is >200 = **then there is an accessory pathway***
- Easy to mistake rapid A fib with an accessory pathway and wide QRS as VT (when caused by a BBB)
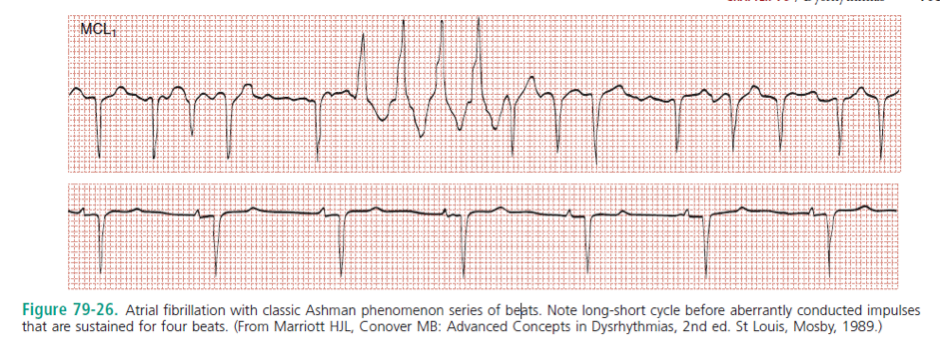
- Ashman phenomenon aberrant Vent. conduction of an early atrial impulse looking like a RBBB and mistaken for VT.
Management:
Highly recommend: https://emergencymedicinecases.com/episode-57-stiell-sessions-2-update-atrial-fibrillation-2014/
Stable:
- AV nodal blocker (CCB/BB) ***unless there is an accessory pathway***, goal HR <110.
- Amiodarone 3-5 mg/kg IV over 15 mins
- Procainamide 18-200 mg/kg is the ONLY safe drug for people with accessory pathways
- need stroke prophylaxis:
Stroke risks (copied information from: https://emergencymedicinecases.com/episode-20-atrial-fibrillation/)
Anticoagulation for Atrial Fibrillation
- Risk of stroke is EQUAL for paroxysmal and persistent A.fib, so even if patient is in NSR at discharge from ED, he or she STILL needs anticoagulation based on risk stratification at least until follow up
- Risk of CVA for non-valvular A.fib is 5% per year (1-2% if <60yo, but 25% if >80yo), and risk reduced to 1% with anticoagulation (risk reduction 70% with NNT 25); risk of CVA with cardioversion is dependent on risk factors and length of A.fib but ranges in 1-5% [in 30 days]
- Use heparin (UFH or LMWH) if patient has arrhythmia of >48hrs or unknown duration and hemodynamically unstable needing emergent cardioversion, or if
Canadian Cardiovascular Society recommends using CHADS2 score to predict risk of CVA
- CHF, HTN, Age≥75, Diabetes, TIA/CVA (2 points) – score ≥1 = dabigatran or warfarin [or DOAC]; consider ASA if score of 1 and patient reluctant to anticoagulate, but urge follow up with GP or cardiologist; also consider ASA if score is 0 and patient is not young
European guidelines recommend using CHA2DS2VASc score given that CHADS2 does not include other (but still important) risk factors
- CHF, HTN, Age ≥75 (2 points), Diabetes, TIA/CVA (2 points), Vascular diseases (CAD, MI, PVD), Age 65-74, Sex category (female 1 point, male 0 point)
- No anticoagulation in younger patients with presumably no structural heart disease and CHA2DS2VASc = 0
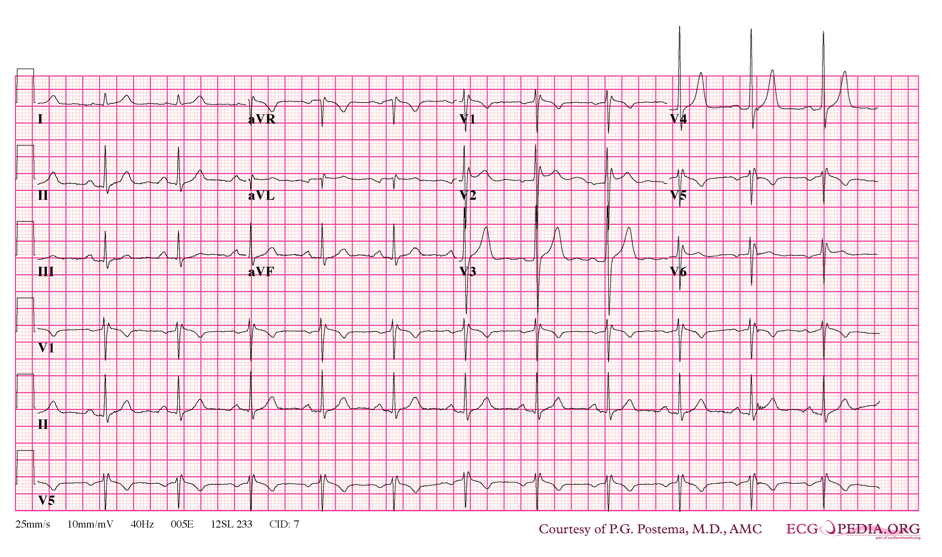
[20] What is Brugada syndrome? What is the management?
BRUGADA SYNDROME (1)
- ventricular dysrhythmia
- sudden death
- syncope
- INHERITED sodium channel disorder
- males in young adulthood
- Dx: ***ecg findings may be transient and only elicited with pharm. stimulation***
- saddleback or coved ST segment in V1-V3
- similar to a RBBB
- Trxt:
- Syncope = ICD
- incidental = referral to cardiologist
WiseCracks:
[1] List 8 side effects of Amiodarone.
[2] Describe features that favour VT over SVT.
[3] Describe the Brugada approach to the diagnosis of VT
“Brugada four-step approach for differentiating ventricular tachycardia (VT) and wide-QRS supraventricular tachycardia (SVT). Only when the response to all four questions is negative is a supraventricular rhythm with abnormal conduction diagnosed. As soon as a single “yes” answer is noted, VT is diagnosed.”
“Only when none of the Brugada criteria are present is a supraventricular cause diagnosed. Although the original investigators found excellent sensitivity (98.7%) and specificity (96.5%) in detecting VT, follow-up investigations have not duplicated this level of accuracy in ED patients (sensitivity 92-94%). Often, physicians either cannot complete the assessment or cannot agree on the findings. In patients receiving class I agents, the Brugada criteria are less reliable.” – Rosen’s 7th Ed.
[4] List Causes of acquired pause-dependent QT prolongation causing Torsades & List causes of adrenergic dependent TdP
TORSADES de pointes
- “twisting of the points”
- polymorphic VT
- Vent rate >200 bpm
- undulating QRS axis with shifting polarity
- paroxysms <90secs
- occurs in the setting of a prolonged QT interval
- may be congenital (triggered by ***FAST HR**) or acquired (***triggered by a slow HR**)
Pause dependent = TdP that is acquired and depends on slow heart rate to trigger the arrhythmia
[5] Describe the treatment of pause dependent TdP
- Common triggers:
- hypoK
- hypoMg
- class 1a and 1c agents
- severe bradyC / AV block
- SAH / MI / CVA /
- congenital
- rare: triggered by adrenergic/tachyC. in children and young adults
- syncope during exertion, Long QT on ECG
- Treatment goal to decrease rate to 100-120
- MgSO4 IV 2-5 g
- Overdrive pacing or beta adrenergic infusion
- Cardioversion
- Acquired Torsades (pause dependent) = speed up the heart
- Congenital Torsades (adrenergic infusion) = slow down the HR with a Beta-blocker
- rare: triggered by adrenergic/tachyC. in children and young adults
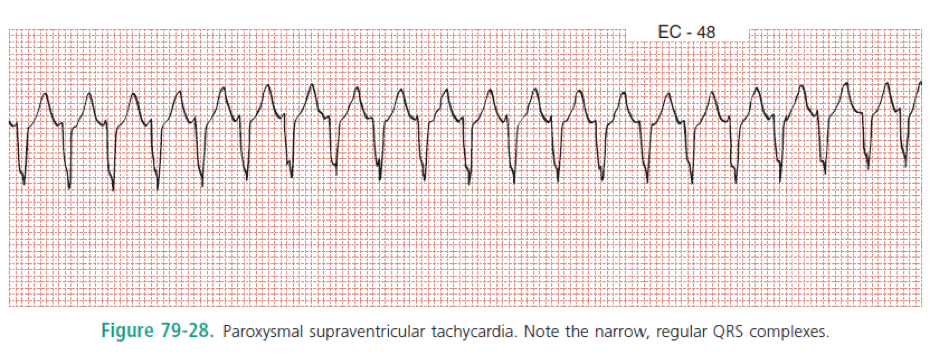
[6] Define PSVT and describe management
“Also known by the less precise term paroxysmal supraventricular tachycardia (PSVT), AVNRT is characterized by a regular narrow-complex rhythm and a ventricular rate of 160 beats/min or greater (Fig. 79-28). It is the most common non sinus tachyarrhythmia in young adults. As its name suggests, AVNRT occurs as the result of a re-entry circuit within the AV node, with normal conduction (narrow QRS) down the bundles of His, and with retrograde conduction (inverted P waves typically buried within the QRS) up into the atria (Fig. 79-29).
The onset of AVNRT is typically abrupt and frequently arises in the context of strenuous exercise or emotional stress. Termination is also abrupt, either spontaneously or following vagal maneuvers that patients are taught or just adopt instinctively. Most patients with AVNRT are symptomatic, but hemodynamic instability is unusual in the absence of underlying cardiopulmonary disease.
If vagal maneuvers fail to restore sinus rhythm, first-line field or ED therapy for AVNRT is adenosine (6 mg rapid large-bore intravenous bolus followed by a flush; repeat with 12 mg if no effect on rate). This approach is successful in 85 to 90% of cases. In refractory cases, diltiazem, esmolol, or metoprolol may be used. Rarely, synchronized cardioversion (at 100-200 J, synchronized and biphasic preferred) is required to terminate AVNRT that is refractory to pharmacologic therapy or in a patient with hemodynamic instability.
Most patients can be discharged once AVNRT has been terminated with adenosine or vagal maneuvers. Patients with frequent recurrences are candidates for prophylaxis (typically with a beta-blocker or calcium channel blocker) or ablation therapy.” – Rosen’s 7th Ed.
- AVNRT
- regular narrow tachyC. >160; usu. a re-entry circuit in the AV node
- abrupt onset
- trxt:
- vagal maneuvers
- adenosine ; if refractory diltiazem or metoprolol / electricity
[7] What is the Ashman phenomenon?
“The Ashman phenomenon refers to aberrant ventricular conduction of an early-arriving atrial impulse following a relatively long R-R interval, as a result of a partially refractory His bundle. Such aberrantly conducted impulses are often seen in atrial fibrillation but can occur in any irregular rhythm in which long-short cycle sequences occur, and they typically assume an RBB block pattern (Fig. 79-26). Ashman beats can be mistaken for PVCs or paroxysmal VT.” – Rosen’s 7th Ed.
[bg_faq_end]This post was uploaded and copyedited by Riley Golby (@RileyJGolby)