This episode of CRACKCast covers Rosen’s 9th Edition Chapter 159, Sedative Hypnotics. While much of the care for overdose is supportive, certain withdrawal patterns can be life-threatening. Knowing how to manage the agitated and sedated patient concurrently are necessary for these presentations.
Shownotes – PDF Here
[bg_faq_start]Key Concepts
- Barbiturate intoxication is rare, and most patients recover with observation and supportive care alone. Hemodialysis is not indicated unless the patient remains hemodynamically unstable despite adequate supportive measures.
- A urine toxicology screen positive for barbiturates does not prove that the drug caused the patient’s clinical condition. Serum barbiturate levels confirm the diagnosis, but do not correlate well with depth of coma or clinical outcome.
- Flumazenil is not indicated in the majority of benzodiazepine overdoses, particularly not in regular benzodiazepine users, in whom flumazenil can precipitate seizures. Because flumazenil’s duration of action (about 1 hour) is much shorter than that of all commonly available benzodiazepines, if flumazenil is used patients should be monitored closely for recurrent respiratory depression or re-sedation.
- Chloral hydrate toxicity may result in sedation and cardiotoxicity, principally in the form of supraventricular tachycardias, which are best treated with a beta blocker.
- Endotracheal intubation to protect against emesis, aspiration pneumonitis, and hypoxia is often necessary for patients with significant CNS or respiratory depression from GHB overdose.
- Withdrawal from GHB or its precursors begins with anxiety, tremor, and insomnia, but it can progress to a severe syndrome characterized by delirium and autonomic instability. Management of this syndrome often requires high-dose benzodiazepine or barbiturate therapy (because they are GABA depleted).
Rosens in Perspective
Big headings for today:
- Barbiturates
- Benzodiazepines
- Special sedatives / OTC sleep aids
- Chloral hydrate
- Gamma-hydroxybutyrate (GHB)
We’ll cover most of these in the core questions, but let’s quickly mention the sleep aids, because we won’t cover them specifically later on.
- Over-the-counter (OTC) sleep aids currently available in the United States contain either diphenhydramine or doxylamine.
- Many preparations also contain acetaminophen or aspirin, added to achieve nighttime pain relief. (check those ASA and acetaminophen levels!!)
- Diphenhydramine and doxylamine are antihistamines that also have hypnotic, antimuscarinic, and weak local anesthetic properties. They act as competitive antagonists of H1 histamine receptors and cause sedation by inhibiting the actions of acetylcholine on muscarinic receptors in the CNS.
- Other special sedatives:
- Flunitrazepam (Rohypnol) – Not sold in the USA, but purchased online and commonly associated as a “date” rape drug
- the Zzzzs
- Zolpidem (Ambien), zaleplon (Sonata), and zopiclone (Imovane) differ in structure from both the benzodiazepines and buspirone, and they are not detected on a benzodiazepine toxicology screen. They act selectively at a specific benzodiazepine receptor, producing sedation without many of the side effects seen with benzodiazepines. They have modest anxiolytic, muscle relaxant, and anticonvulsant properties.
- Management is supportive!
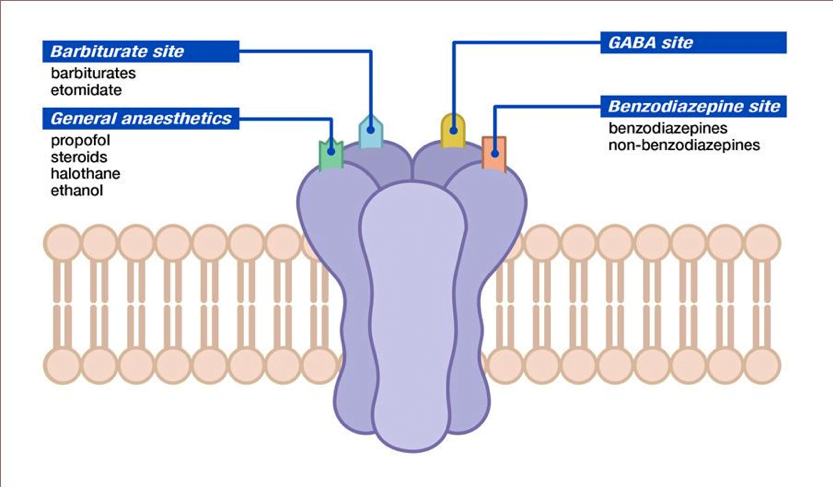
[1] Describe the components of the GABA receptor complex and their physiologic effect
- The GABA receptor is a protein complex found on postsynaptic membranes in the CNS
- Structurally, it consists of several distinct receptor sites surrounding a chloride ion (Cl−) channel (Fig. 159.1).
- GABA opens the chloride channel. The resulting flow of Cl− into the cell increases the negative resting potential, hyperpolarizing and stabilizing the membrane.
- There are separate receptor sites for barbiturates and for benzodiazepines and a third site that binds GABA, ethanol, and meprobamate.
- Although barbiturates and ethanol can directly increase Cl− conductance, benzodiazepines require the presence of GABA to affect Cl− flow, which may account for the relative safety of benzodiazepines in comparison with barbiturates.
[2] Describe the sedative-hypnotic toxidrome, and list 6 drugs in the ddx.
Toxidrome
- Depressed HR / RR
- No pupil changes
- Possibly dilated
- Possible nystagmus
Ddx
- Barbiturates
- Benzodiazepines
- Zolpidem
- Chloral Hydrate
- OTC sleep aids (benadryl)
- Gamma Hydroxybutyrate (GHB)
[3] How do barbiturates work?
- Barbiturates depress the activity of all excitable cells, especially central nervous system (CNS), enhancing the activity of gamma-aminobutyric acid (GABA)
- In acute overdose decrease neural transmission in autonomic ganglia, the myocardium, and the gastrointestinal tract and also inhibit the response to acetylcholine at the neuromuscular junction.
- Barbiturates produce dose-related depressive effects = mild sedation < coma < fatal respiratory arrest.
- Laryngospasm can occur at low doses
- Large doses can cause delaying of gastric emptying
- Therapeutic doses = similar to sleep
- In overdose, there are other effects
- Early stages = some patients experience euphoria / allodynia
- Later -> Act directly on the medulla = respiratory depression.
- With toxic doses, more significant hypotension occurs from direct depression of the myocardium along with pooling of blood in a dilated venous system.
- Barbiturates decrease cerebral blood flow and intracerebral pressure.
- Barbiturates are classified according to their onset and duration of action
- Only long-acting preparations have anticonvulsant effects in doses that do not cause sedation.
- Short- and intermediate-acting preparations are almost completely metabolized to inactive metabolites in the liver, whereas 25% of a phenobarbital (long-acting) dose is excreted unchanged through the kidney.
- Barbiturates cross the placenta with fetal levels approaching those of the mother. They are also excreted in low concentration in breast milk. Use during pregnancy is associated with birth defects (category D).
[4] What are the clinical symptoms of barbiturate overdose?
Mild toxicity
- Drowsiness
- slurred speech
- Ataxia
- unsteady gait
- Nystagmus
- emotional lability
- impaired cognition
Severe Toxicity
- CNS depression = stupor to deep coma and respiratory arrest
- Pupils are usually normal or small and reactive
- hypoxia can cause pupils to be fixed and dilated
- Corneal and gag reflexes may be diminished or absent
- muscle tone = flaccid
- DTRs = diminished or absent
- Flexor (decorticate) and extensor (decerebrate) posturing can occur in patients comatose from barbiturate intoxication
- DON’T PRONOUNCE SOMEONE BRAIN DEAD IF THEY MAY HAVE OVERDOSED ON A BARBITURATE = Even their EEG will look like brain death
- Depressed gastrointestinal motility = delaying drug absorption
- As the drug is metabolized and blood levels drop, peristalsis and drug absorption may increase, causing drug levels to rise again!!
- Life threat = respiratory depression
- Hypotension w/ normal or increased heart rate.
- Watch out for noncardiogenic pulmonary edema & aspiration pneumonitis
***Note: Barbiturate withdrawal syndrome includes:
- Tremors
- Hallucinations
- Seizures
- delirium (similar to the delirium tremens of ethanol withdrawal)
[5] How are barbiturate overdoses managed?
Investigations / Supportive care / Invasive ventilation as needed
Labs:
- The therapeutic level of phenobarbital is 15 to 40 μg/mL (65 to 172 μmol/L)
- A serum level greater than 50 μg/mL can be associated with coma, especially in a patient who is not a chronic user.
- Levels greater than 80 μg/mL cause potentially fatal respiratory depression.
- Serial phenobarbital levels can monitor effectiveness of treatment. Other than phenobarbital, barbiturates have high volumes of distribution, so serum levels do not accurately re ect CNS concentrations or correlate with clinical severity.
- A positive urine screen establishes only qualitative exposure to a barbiturate but does not prove that the drug is present in toxic amounts and should not be relied on to explain decreased mental status.
- CXR = look for pulmonary edema or aspiration
- Consider neuroimaging +/- EEG to rule out non-convulsive status
Management
- ABCs
- Intubate as needed
- Fluids +/- pressors for hypotension (watch out for pulmonary edema)
- Decontamination
- Potentially MDAC (but no evidence for improved outcome)
- Consult your local toxicologist
- Enhanced Elimination
- Urine Alkalinization = controversial
- Hemodialysis
- serum levels over 100 mcg/mL
- refractory hypotension
- renal or cardiac failure
- acid-base or electrolyte abnormalities
- inadequate response to less invasive measures
Disposition
- Observe 6 hrs
- Usually symptomatic by 1 hr
- If they are sick = ICU
[6] How do benzodiazepines work?
- Punch line = work by enhancing the inhibitory actions of GABA
- Specific binding site potentiates GABA effects on the chloride channel= intracellular flux of chloride ions and hyperpolarizing the cell
- The net effect is a diminished ability of the nerve cell to initiate an action potential, inhibiting neural transmission
- Three unique benzodiazepine receptors exist:
- distributed throughout CNS / PNS
[7] List risk factors for benzodiazepine withdrawal and its management.
Risk Factors:
- Abrupt discontinuation
- Chronic user
- Dose of benzodiazepine and the duration of its use.
- Continuous treatment for more than 4 months is generally required before a patient is at risk for withdrawal.
Management
- Treatment of withdrawal = restarting benzodiazepines.
Benzodiazepine Withdrawal Symptoms
- Non-specific
- Anxiety, depression, insomnia, tremor, tachycardia, sweating
- Severe (rare)
- Visual hallucinations, delirium, seizures
[8] What are the indications for flumazenil? What are the contraindications? (box 159.2)
Indications
- Isolated benzodiazepine overdose in non-habituated user (eg. Accidental pediatric)
- Reversal of procedural sedation
Absolute Contraindications
- Suspected co-ingestion lowering seizure threshold (see box 159.2)
- Patient taking medicine to control life threatening condition (eg. Seizures)
- Concurrent sedative-hypnotic withdrawal
- Seizure activity or myoclonus
- Hypersensitivity to flumazenil or benzodiazepines
- Patient with muscular blockade
Relative Contraindications
- Chronic benzo use not taken for controlling life threatening conditions
- Known seizure disorder not treated with benzos
- Head injury
- Panic attacks
- Chronic alcoholism
[9] How does chloral hydrate toxicity present?
- CNS and respiratory depression
- Gastrointestinal irritation
- Cardiovascular instability, and dysrhythmias
- decreases myocardial contractility, shortens the cardiac refractory period, and increases the sensitivity of myocardium to catecholamines.
- AFIB/ SVT / VT/ multifocal PVCs / torsades de pointes, VFIB & asystole
- The combination of deep coma and cardiac dysrhythmia without hypoxia is characteristic of severe cases.
- Classically described as “A pear-like odor on breath or gastric contents”
Findings consistent with chloral hydrate toxicity include:
- Miosis
- Muscle flaccidity & diminished DTRs
- Hypoventilation
- Hypotension
- hypothermia
Other Details
- Chloral hydrate = corrosive
- Watch for GI irritation or damage = V/D, esophagitis, hemorrhagic gastritis, and, rarely, gastrointestinal perforation or necrosis
- Transient hepatic or renal dysfunction can also occur.
Dosing Danger
- The hypnotic oral adult dose=0.5 to 1.0 g.
- The toxic oral dose = ~ 10 g in adults
- may be as little as 1.5 g in children.
- Onset of action is 20 to 30 minutes.
- Chloral hydrate and ethanol in combination (historically referred to as a “Mickey Finn”) potentiate each other’s action to produce rapid loss of consciousness. Chloral hydrate increases the half-life of ethanol by competitively inhibiting the enzyme alcohol dehydrogenase, and the metabolism of ethanol generates nicotinamide adenine dinucleotide (NADH), a cofactor for the conversion of chloral hydrate to trichloroethanol.
[10] What is the clinical presentation of GHB toxicity?
- Diagnosis = history and clinical course
- Classically seen as :
- Rapid recovery from coma, or
- Periods of agitation alternating with periods of decreased level of consciousness (binds to both GHB receptors and GABAB receptors (excitatory))
- Classically come in being bagged by EMS, wake up with a bang and start swinging!
- Hypothermia may occur with prolonged coma
- You may see bradycardia with or without hypotension
- Miosis with or without nystagmus may be seen.
- 50% of GHB overdoses WILL VOMIT = aspiration pneumonitis
- Don’t confuse random myoclonic movements with seizures
[11] How is GHB withdrawal managed?
“Patients who suddenly stop GHB or its precursors after chronic, frequent use can experience a severe and potentially life- threatening withdrawal syndrome.” – Rosen’s
- GHB has a short half-life = symptoms of withdrawal begin within several hours of the last dose.
- Typically users consume GHB every 1 to 3 hours 24/7 to avoid withdrawal symptoms
Mild withdrawal involves anxiety, tremor, and insomnia. This can progress to severe with confusion, delirium, overt psychosis, paranoid ideation, hallucinations (visual, aural, or tactile), and autonomic instability.
Treatment
- Initial = high-dose benzodiazepines (lorazepam 1 to 2 mg IVP Q30 to 60 minutes
- NOTE***GHB withdrawal may =depleted levels of GABA.
- Benzos require GABA to be present to work
- Thus they may not be effective in control of GHB withdrawal
- So… Barbiturates are the way to go ( they do not need GABA to be effective)
- Watch for rhabdomyolysis or hyperthermia
Wisecracks
[bg_faq_start][1] List 2 drugs used for ‘date-rape’ and describe their toxicity
- GHB
- Psychoactive agent
- GHB receptor
- Like ETOH
- Mix picture = decreased LOC alternating with aggression
- Flunitrazepam (Rohypnol)
- Benzodiazepine (potent)
- GHB & GABA receptor
[2] What Benzos WILL NOT be detected on urine drug screen?
Basically comes down to these drugs not being metabolised to oxazepam glucuronide
- Clonazepam
- Lorazepam
- Midazolam
- Alprazolam
- Flunitrazepam
This post was uploaded and copyedited by Riley Golby (@RileyJGolby)